A recent study using advanced protein structure modeling has more than tripled the number of known eukaryotic-like proteins in Asgard archaea, a group of microbes considered the closest living relatives to the ancestor of all complex life. This discovery suggests the ancient archaeal ancestor of eukaryotes was significantly more complex than previously understood, possessing building blocks for cellular structures once thought to be unique to more advanced organisms.
Key Takeaways
- Researchers identified 908 new eukaryotic-like proteins in Asgard archaea using protein structure modeling, a method more sensitive than traditional sequence comparison.
- These newly found proteins, termed 'isomorphic ESPs' (iESPs), expand the known repertoire of eukaryotic features in these ancient microbes.
- The findings include components for complex cellular machinery like the Vault and Commander complexes, which are involved in transport and processing within eukaryotic cells.
- This research challenges previous assumptions about the cellular simplicity of the archaeal ancestor that gave rise to all eukaryotes, including plants, animals, and fungi.
A New Window into Deep Evolution
The origin of the eukaryotic cell, which contains a nucleus and other membrane-bound organelles, represents a major evolutionary leap. For decades, scientists have worked to piece together the puzzle of how these complex cells emerged from simpler prokaryotic ancestors approximately two billion years ago.
Recent discoveries identified a group of microbes called Asgard archaea as the closest known relatives to the original archaeal host cell in this transition. Their genomes contain genes for proteins previously thought to be exclusive to eukaryotes, known as Eukaryotic Signature Proteins (ESPs).
However, detecting these ancient connections is difficult. Over vast evolutionary time, the genetic sequences of proteins can change so much that they become unrecognizable, even if their three-dimensional structure and function remain similar. This is often referred to as the 'twilight zone' of sequence similarity, where traditional comparison methods fail.
What are Asgard Archaea?
Asgard archaea are a superphylum of single-celled microorganisms found in deep-sea sediments and other extreme environments. They are significant because their genomes contain more eukaryotic-like genes than any other known prokaryote, positioning them as a crucial link in understanding the origin of complex life.
Beyond Sequence: The Power of Structural Modeling
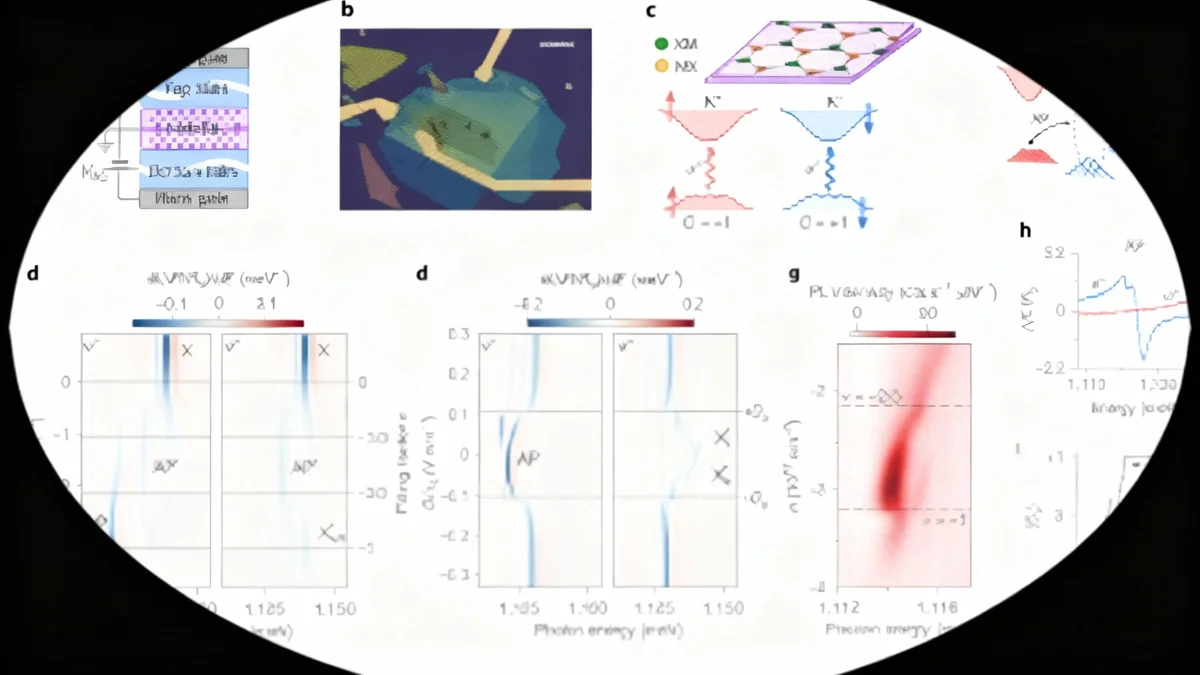
To overcome the limitations of sequence-based analysis, a team of researchers employed advanced computational tools like AlphaFold and ESMfold. These programs predict the 3D structure of a protein from its amino acid sequence, allowing for comparisons based on shape rather than just the underlying genetic code.
The team analyzed a massive dataset of 936 Asgard archaeal genomes, generating structural models for over 37,000 representative proteins. By comparing these structures to a database of known eukaryotic proteins, they could identify matches that were invisible to conventional methods.
This structural approach led to the identification of 908 new structure-based ESPs, which the researchers termed 'isomorphic ESPs' or iESPs. This number more than triples the previously known count of such proteins in Asgard archaea, dramatically expanding the known toolkit of our ancient microbial ancestors.
From 1,323 to Over 3,500
Prior to this study, approximately 1,323 ESPs were identified in Asgard archaea. The addition of 908 new iESPs and the re-categorization of existing ones brings the total number of potential ancestral proteins to a much higher figure, fundamentally changing our view of the archaeal ancestor's complexity.
Discovering Eukaryotic Machinery in an Archaeon
The newly identified iESPs are not just random proteins; they include components of sophisticated cellular machinery crucial for eukaryotic life. Among the most significant finds are proteins structurally similar to key parts of two major eukaryotic complexes.
The Vault Complex
Researchers discovered Asgard archaeal proteins that are structural matches for the Major Vault Protein (MVP), the primary component of the vault, the largest known ribonucleoprotein complex in eukaryotes. In humans, vaults are thought to be involved in cellular transport and stress response.
The identification of an MVP homologue in Asgard archaea suggests a prokaryotic origin for this complex cellular structure, which was previously thought to have no clear ancestor outside of eukaryotes.
Structural modeling suggests that ten of these Asgard MVP-like proteins could self-assemble into a cup-like structure, reminiscent of the eukaryotic vault's architecture.
The Commander Complex
Another key discovery was an Asgard archaeal protein with a structure matching that of COMMD proteins. In eukaryotes, COMMD proteins form part of the Commander complex, which is essential for recycling materials from a cellular sorting station called the endosome.
Despite having an extremely low sequence identity of only 8.5% with its eukaryotic counterpart, the 3D structure was a clear match. This highlights the power of the structural approach to find deeply hidden evolutionary links.
The study also identified Asgard homologues for other important eukaryotic proteins, including:
- Ufm1 (Ubiquitin fold modifier 1): A protein involved in stress responses within the cell, previously found only in eukaryotes.
- CINP (Cyclin-dependent kinase 2-interacting protein): A protein involved in DNA replication and damage control.
A More Complex Ancestor
These findings collectively paint a picture of the archaeal ancestor of eukaryotes as being far more sophisticated than previously imagined. It wasn't just a simple cell that later acquired complexity; it already possessed a rich collection of proteins that would become the foundation for eukaryotic cellular organization.
The newly identified iESPs are involved in a wide range of functions. While previous studies found many ESPs related to the cytoskeleton and intracellular trafficking, this new research uncovered a large number involved in information storage and processing (like translation and ribosome biogenesis) and metabolism.
Metabolic Surprises
The study found 137 iESP clusters related to metabolism, compared to just 24 previously known ESPs in this category. This suggests that the archaeal contribution to eukaryotic metabolism may be significantly underestimated.
While the exact function of these proteins in modern Asgard archaea remains to be confirmed through experimental studies, their structural similarity to eukaryotic proteins provides strong evidence of a shared evolutionary history. This research opens new avenues for understanding the step-by-step process that led to the emergence of complex life on Earth.
Future work will likely focus on biochemically characterizing these Asgard proteins to determine if they function in a similar manner to their eukaryotic relatives. Such studies will be crucial for refining our models of one of the most important transitions in the history of life.